Table of Contents=> Go Directly
ATOMIC THEORY
An Atomic theory is a model developed to explain the properties and behaviors of atoms. As with any scientific theory, an atomic theory is based on scientific evidence available at any given time and serves to suggest future lines of research about atoms.
The concept of an atom can be traced to debates among Greek philosophers that took place around the sixth century B.C. One of the questions that interested these thinkers was the nature of matter. Is matter, they asked, continuous or discontinuous? That is, if you could break apart a piece of chalk as long as you wanted, would you ever reach some ultimate particle beyond which further division was impossible? Or could you keep up that process of division forever? A proponent of the ultimate particle concept was the philosopher Democritus (c. 470–c. 380 B.C. ), who named those particles atomos. In Greek, atomos means “indivisible.”

Ancient Greek Beliefs
Leucippus and Democritus were the first to propose, in the 15th century B.C., that all matter is made of tiny units called atoms. The two philosophers held that these were solid particles without internal structure, and came in a variety of shapes and sizes. Intangible qualities such as taste and color, according to this theory, were made of atoms. However, Aristotle strongly opposed this idea, and the scientific community failed to pay serious attention to it for centuries.

The debate over ultimate particles was never resolved. Greek philosophers had no interest in testing their ideas with experiments. They preferred to choose those concepts that were most sound logically. For more than 2,000 years, the Democritus concept of atoms languished as kind of a secondary interest among scientists.
Then, in the first decade of the 1800s, the idea was revived. English chemist John Dalton (1766–1844) proposed the first modern atomic theory. Dalton’s
theory can be called modern because it contained statements about atoms that could be tested experimentally. Dalton’s theory had five major parts. He said:
All matter is composed of very small particles called atoms.
All atoms of a given element are identical.
Atoms cannot be created, destroyed, or subdivided.
In chemical reactions, atoms combine with or separate from other atoms.
In chemical reactions, atoms combine with each other in simple, whole-number ratios to form combined atoms.
Dalton’s atomic theory is important not because everything he said was correct. It wasn’t. Instead, its value lies in the research ideas it contains. As you read through the list above, you’ll see that every idea can be tested by experiment.

English physicist J.J Thomson
proposed the “plum pudding” theory of the divisible atom in 1904, after discovering electrons in 1897. His model postulated that atoms consist of a big positively-charged sphere studded with negatively charged electrons (he called them “corpuscles”) like fruit in a plum pudding. He further hypothesized that the charge of the positive sphere’s charge is equal to the negative charges of the electrons. Today we call the positive charged particles protons, and the negative ones electrons.
British physicist Ernest Rutherford proposed a nuclear model of the atom, in which a nucleus exists, in 1911. He also discovered activity in this part, namely the movement of protons and electrons within the central part of the atom. He further postulated that the number of protons in an atom equals that of the electrons.J.J Thomson
also hypothesized that more neutral particles exist. These have come to be known as neutrons.
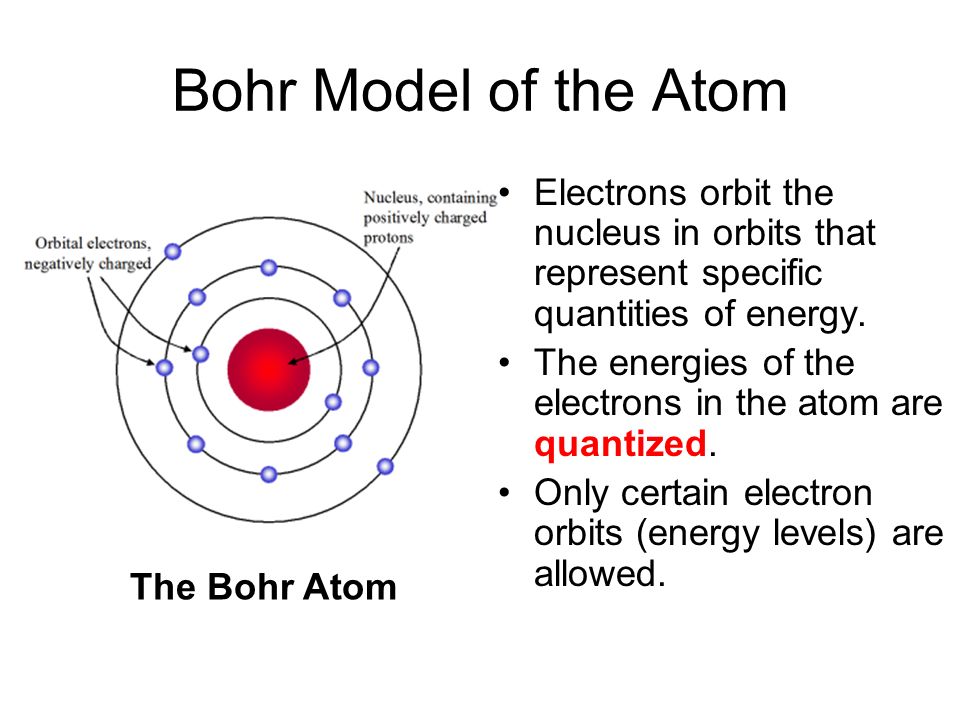
In 1913 Bohr proposed his quantize shell model of the atom to explain how electrons can have stable orbits around the nucleus. The motion of the electrons in the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the electrons would lose energy and spiral into the nucleus. To remedy the stability problem, Bohr modified the Rutherford model by requiring that the electrons move in orbits of fixed size and energy. The energy of an electron depends on the size of the orbit and is lower for smaller orbits. Radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no orbit of lower energy into which the electron can jump.
From decades of painstaking research from thousands of scientists, the current atomic theory builds on work done in the 1930s by Albert Einstein, Werner Heisenberg and others. As with the earlier theories, the atom consists of a central, heavy nucleus surrounded by a number of electrons. Unlike earlier theories that treated electrons, protons and other tiny particles as definite solid “lumps,” modern quantum theory treats them as statistical “clouds;” oddly, you can measure their speed exactly, or their locations, but not both at the same time. Instead of electrons behaving as planets orbiting in well-behaved elliptical paths, they whirl around in fuzzy clouds of various shapes. Atoms, then become less like hard, precise billiard balls and more like springy, round sponges. And despite being “solid” matter, they can exhibit wavelike properties such as wave length and interference patterns.
Quarks have fractional element charges. There are six identified varieties of quarks:

The Up and Down Quarks
The Up and Down quarks are the most common quarks and weigh the least out of all six flavors. They are identified as making up most ordinary matter, such as protons and nuetrons.
The Strange Quark
Just like a typical science-tale, the strange quark was discovered out of complete “strangeness”. During a study of cosmic ray interactions, in 1947, a product of a proton collision with a nucleus was found to live for a much longer time than what was hypothesized.
The Charm Quark
Originally discovered as the J/Psi Particle (a meson) in 1974, this newly founded quark had over three times the mass of a proton, weighing in at about 3100 MeV. This particle decayed slowly and did not fit into the framwork of the up, down and strange quarks. This particle became the first firm experimental evidence for the fourth quark, the Charm Quark
The Bottom Quark
In 1977, an experimental group from Fermi lab discovered quark-antiquary pair at 9.4 GeV/c2, which was interpreted at the bottom-anti bottom quark and was called the Upsilon Meson. The Upsilon particle was the first evidence of the existence of the fifth quark from the 1977 Fermi lab experiment. From this experiment, the mass of the bottom quark results to be about 5 GeV/c2.
The Top Quark
In April 1995, evidence of the existence of the Top Quark was proven. The evidence was found in the collision products of 9.0 TeV protons with equally energetic anti protons (anti quarks are explained in section 5) in the proton-anti proton collider. The evidence involved analyzing trillions of proton-anti proton collisions. These trillions of proton-anti proton collisions were categorized into two groups. The first group is known as the Collider Detector Facility group and the second group is the D-0 group.
The Anti quark
Just like love and marriage, quarks do not exist alone. They are bound in quark-anti quark pairs or triplets by the strong force. Every quark carries some charge and its anti quark carries the opposite valued-charge that the quark maintains. However, for charge zero mesons with different types of quarks and anti quarks, there is an antiparticle that reverses the role and quark and anti quark.

This model of the atom can be used to explain many of the ideas in chemistry in which ordinary people are interested. But the model has not been used by chemists themselves for many decades. The reason for this difference is that revolutionary changes occurred in physics during the 1920s. These changes included the rise of relativity, quantum theory, and uncertainty that forced chemists to rethink the most basic concepts about atoms.
As an example, the principle of uncertainty says that it is impossible to describe with perfect accuracy both the position and the motion of an object. In other words, you might be able to say very accurately where an electron is located in an atom, but to do so reduces the accuracy with which you can describe its motion.
By the end of the 1920s, then, chemists had begun to look for new ways to describe the atom that would incorporate the new discoveries in physics. One step in this direction was to rely less on physical models and more on mathematical models. That is, chemists began to give up on the idea of an electron as a tiny particle carrying an electrical charge traveling in a certain direction with a certain speed in a certain part of an atom. Instead, they began to look for mathematical equations which, when solved, gave the correct answers for the charge, mass, speed, spin, and other properties of the electron.
Related Links
Science
Physics
Chemistry
Biology
Geography
Scientists
